Glossary
Alloy. An alloy is made by combining metal elements to improve the strength, durability, color, or melting point of a single metal element. It can be a less valuable base metal combination, such as brass or bronze, or a more valuable precious metal combination, such as sterling silver and karat gold.
Annealing. A multistep process of heating and stressing metal to improve its strength and hardness.
Anodization. An electrochemical process that permanently alters the surface of nonferrous metals, making them corrosion resistant. Aluminum is frequently anodized.
Base metal. Base metals — copper, iron, and nickel — corrode or oxidize easily. Base metal is also a generic term used to describe all metals and alloys that are not otherwise classified as precious metals.
Corrosion. The process of deterioration that occurs when certain metals are exposed to water or oxygen.
Draw plate. A hardened, steel plate pierced with a number of holes in decreasing sizes. Wire is pulled through successively smaller holes on the draw plate to make it thinner.
Ductility. The physical quality of a metal that allows it to be drawn or stretched without cracking or breaking.
Fusing. To mix metals together by melting.
Gauge. Gauge is a numerical representation of the diameter of round wire or the cross-sectional dimension of shaped wire.
Hypoallergenic. Metals that tend to cause fewer allergic skin reactions than other metals.
Karat. A numerical representation of the percentage of pure gold found in a gold alloy. Karat is not to be confused with carat, a term that refers to the weight of diamonds and other precious gemstones.
Luster. The way light reflects off the surface of metal, causing it to glow.
Malleability. The physical quality of a metal that allows it to be manipulated, compressed, hammered, and rolled.
Ounce. A measurement of weight most commonly used in the United States. There are 16 ounces in 1 pound.
Pennyweight. A unit of measure equal to 1/ 20 troy ounce. In the Middle Ages, money was based on the weight of sterling silver. A British penny was 1/ 20 ounce or 1/ 240 pound of sterling silver.
Plating or electroplating. The process of applying a thin layer of precious metal or alloy to the surface of a conductive material, namely another metal, with electricity.
Tarnish. A thin layer of corrosion that forms over certain metals when they are exposed to air, causing the metal to darken and lose its luster.
Temper. The hardness or softness of a particular wire. Tempers range from dead-soft to spring-hard.
Troy ounce. An imperial measurement of weight. There are about 32 troy ounces in 1 kilogram.
Appendix
Types of Metal
The type of metal chosen for a jewelry project is largely a matter of personal preference. Things to consider when selecting a metal are cost, workability, and, of course, color. Precious metals are the most sought after, but they are also the most costly. Metal alloys and precious metal-plated base metals are attractive and cost-effective alternatives.
By definition, the terms precious and base are chemical references that describe the corrosive qualities of different metals. In the marketplace, these terms are used to categorize metals by their rarity and their value. The precious metals, also known as noble metals, that are desirable for jewelry making include gold, silver, and platinum. These metals are highly resistant to corrosion, though silver will tarnish. Precious metals are easier to work with because they are more malleable and they have a naturally shinier luster than other metals. Precious metals are also commodities, and their cost fluctuates daily with the world markets.
Base metal elements, including iron, nickel, and copper, occur naturally in large quantities. The abundance of these elements makes wire and metal components made from base metal alloys far less expensive than precious metals. Base metal is also a generic term used to describe all metals used in jewelry making that are not classified as precious metals. Base metal alloys include pewter, brass, and bronze.
GOLD
Gold (Au) has been popular for its rarity and beauty since before written history. Gold is the most malleable of all metals, making it the easiest to work with. Because pure gold is generally too soft to work with when making wire, chain, and jewelry findings, it is mixed with other metals, including copper and zinc, to produce karat gold. The term karat refers to the percentage of pure gold in popular karat weights, which ranges from 10K to 24k (see table below).
Gold-filled (G/F) is a process by which 14K or 18K gold is bonded to a base metal, usually brass but sometimes copper. Generally 5% of the total weight and the entire surface of a gold-filled item is karat gold.
Gold-plate (G/P) is a process by which a thin layer of karat gold is electroplated to a base metal like brass or steel. The gold-plated layer is thinner than the layer applied in gold-filled wire and will wear off over time.
Vermeil is a process by which 24K or 18K, but no less than 14K gold, is flash-plated or micron-plated to a sterling silver or fine silver core. Flash-plating involves dipping or washing a silver core in electroplating solution for a very short period of time. The gold surface of flash-plated vermeil can wear off in a matter of days. Micron-plating involves soaking a silver core in electroplating solution for an extended period of time until a 1 to 2.5 micron layer of gold has coated the surface. The gold surface of micron-plated vermeil will also wear off in time.
White gold is an alloy of gold and white metals like zinc, nickel, or silver. Even though white gold is tarnish resistant, it is brittle and usually needs to be rhodium-plated. White gold is a cost-effective alternative to platinum but can cause allergic reactions once the plating wears off.
Rose gold is an alloy of gold and copper that results in a golden metal with a reddish or pinkish hue. Typically, the ratio of gold to copper is 3 to 1, but other percentages can be found. The lightness or darkness of the rose hue results from the amount of copper added. Rose gold will patina over time.
Rose gold–filled is a process that permanently bonds a layer of rose gold alloy to a base metal alloy like copper, nickel, or brass.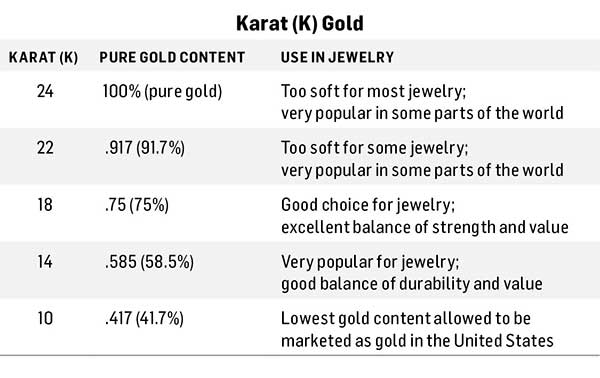
SILVER
Silver (Ag) is a very soft, easily formed, silvery white metal. Oxidation will cause silver to darken or tarnish over time. Silver is frequently mixed with other metals to make it more durable for use in jewelry-making components.
Fine silver has a .999 level of purity. Fine silver is the purest and softest silver available. Thai silver beads and findings, which are often elaborately stamped, are made from fine silver.
Sterling silver has a .925 level of purity. This silver alloy is made from 92.5% pure silver and 7.5% copper. True sterling silver will always be stamped “.925.”
Argentium is a modern sterling silver alloy that contains 92.5% pure silver and 7.5% copper and germanium. Germanium gives this alloy increased tarnish resistance and prevents fire scale. These properties make Argentium a very desirable, though expensive, metal.
Silver-filled is a process by which fine silver or sterling silver is bonded to a base metal alloy like nickel or brass. The total weight of a silver-filled item is notated as either 1/ 10 (5%) or 1/ 20 (10%) fine silver or sterling silver.
Silver-plate is a process by which a thin layer of fine silver or sterling silver is electroplated to a base metal like brass or steel. The silver-plated layer is thinner than the silver-filled layer and will wear off over time.
OTHER METALS
Nickel (Ni) looks like silver but can cause a skin reaction in many people. It is a tough, corrosion-resistant material that is easily molded and often used as an element is metal alloys.
Nickel silver (also known as German silver or gun metal) is not silver at all. It is a silvery, hard, corrosion-resistant, malleable alloy of 65% copper, 18% zinc, and 17% nickel.
Stainless steel is a metal alloy that contains 90% steel and 10% chromium. Since it does not corrode, rust, or tarnish, it is an attractive metal for jewelry-making components.
Surgical steel (SSS) is a medical-grade, corrosion-resistant, and rust-resistant stainless steel that is hypoallergenic.
Pewter is a malleable, silver-toned alloy containing 90% tin and 10% copper. The addition of copper is used to harden the alloy. Low-quality pewter, with a bluish tint, is hardened with lead, which is considered to be carcinogenic. Pewter has a very low melting point that makes it ideal for casting.
Britannia is a pewter alloy containing 92% tin, 6% antimony, and 2% copper. It is essentially lead and cadmium free.
Copper (Cu) is the oldest-known metal and is often associated with having healing powers. It is a reddish-brown, metallic element that patinas to a warm brown. Copper can also take on a green patina with oxidization. Copper is often an element in metal alloys.
Bronze (Bz) is an alloy of 90% copper and 10% tin.
Brass (Br) is an alloy of 75% copper and 25% zinc that is gold in color. Brass will tarnish and turn brown over time.
Niobium (Nb) is a soft gray metal with very low toxicity, which makes it hypoallergenic.
Don’t Bead without These Books!
READ MORE AT STOREY.COM
You can read the previous entry (Chapter Five) in the series here.

